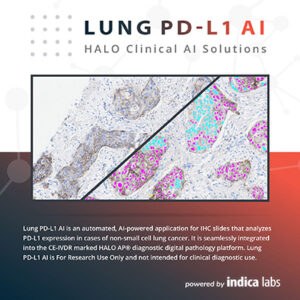
Lung PD-L1 AI is an automated, AI-based decision-support tool that standardizes scoring of PD-L1 IHC in non-small cell lung cancer (NSCLC) on whole slide images (WSI) in accordance with clinical scoring guidelines. Lung PD-L1 AI is seamlessly integrated into the HALO AP® diagnostic digital pathology platform.
Intended Use
Lung PD-L1 AI is For Research Use Only and not intended for clinical diagnostic use.
Inputs
Lung resections and core biopsies
Key Outputs
Tumor Proportion Score (TPS)
Supported Clones
SP263, 22C3
Lung PD-L1 AI SP263 clone validation was performed using a 3D Histech P1000 scanner. 22C3 clone validation was performed using a Leica GT450 scanner.

Automated PD-L1 Tumor Proportion Scoring Algorithm in Non-Small Cell Lung Cancer for Multiple Companion Diagnostic Assays
Read about the development and validation of an AI-based PD-L1 scoring algorithm for HALO AP®.
File Formats
- Non-proprietary (JPG, TIF, OME. TIFF, DICOM [DCM*])
- Leica (SVS, AFI, SCN, LIF)
- Hamamatsu (NDPI, NDPIS)
- Philips (iSyntax, i2Syntax)
- 3DHistech (MRXS)
- Nikon (ND2)
- Akoya (QPTIFF, component TIFF)
- Olympus / Evident (VSI)
- Zeiss (CZI)
- Ventana (BIF)
- KFBIO (KFB, KFBF)
*whole slide images
Lung PD-L1 AI Brochure
Learn more about the automated workflow and benefits of utilizing Lung PD-L1 AI.

Automated PD-L1 Tumor Proportion Scoring Algorithm in Non-Small Cell Lung Cancer for Multiple Companion Diagnostic Assays
Read about the development and validation of an AI-based PD-L1 scoring algorithm for HALO AP®.

Standardize PD-L1 Evaluation
Standardization facilitates comparison across individuals, laboratories, and institutes, which in turn enables collaborative research, enhances data reliability, and ultimately advances scientific understanding.

Increase Your Workflow Efficiency
Lung PD-L1 AI can assist pathologists and researchers in conducting more efficient PD-L1 expression evaluation. The algorithm can be run on slides before they are released for review, so you can make faster and more informed decisions.

Complement Your Expertise
Lung PD-L1 AI complements your expertise and provides an easy-to-read visual mask that highlights tumor areas and positive and negative cells with colorimetric feedback. Analysis results include cell-level information, informing your PD-L1 IHC evaluation process.
Comprehensive Analysis
Lung PD-L1 AI assay reports a comprehensive set of results, including TPS score, total cell count, number of tumor cells, and number of PD-L1 positive and negative tumor cells. The Lung PD-L1 AI algorithm includes built-in and validated AI-based exclusion of artifact and benign epithelial regions with markups that can be verified by the pathologist, so you can be certain that analysis is performed accurately and consistently each and every time.
Seamless Deployment in HALO AP®
Lung PD-L1 AI is deployed and fully integrated into HALO AP®, the AI-powered, pathologist-driven platform for anatomic pathology workflows from Indica Labs.
Want to Learn More?
Fill out the form below to request a live demo of Lung PD-L1 AI or to learn more about our other clinical solutions.
You can also drop us an email at info@indicalab.com
Products & Services
Interested in purchasing or learning more about our products and services? Our highly trained application scientists are a couple of clicks away.
Software Maintenance & Support Coverage
Interested in purchasing an SMS plan? We would be happy to give you a quote.
Technical Support
Need technical support? Our IT specialists are here to help.
Lung PD-L1 AI is For Research Use Only and not intended for clinical diagnostic use. Lung PD-L1 AI is accessed via the HALO AP® enterprise digital pathology platform.
HALO AP® is CE-IVDR marked for in-vitro diagnostic use in Europe, the UK, and Switzerland. HALO AP is For Research Use Only in the USA and is not FDA cleared for clinical diagnostic use. In addition, HALO AP provides built-in compliance with FDA 21 CFR Part 11, HIPAA, and GDPR.