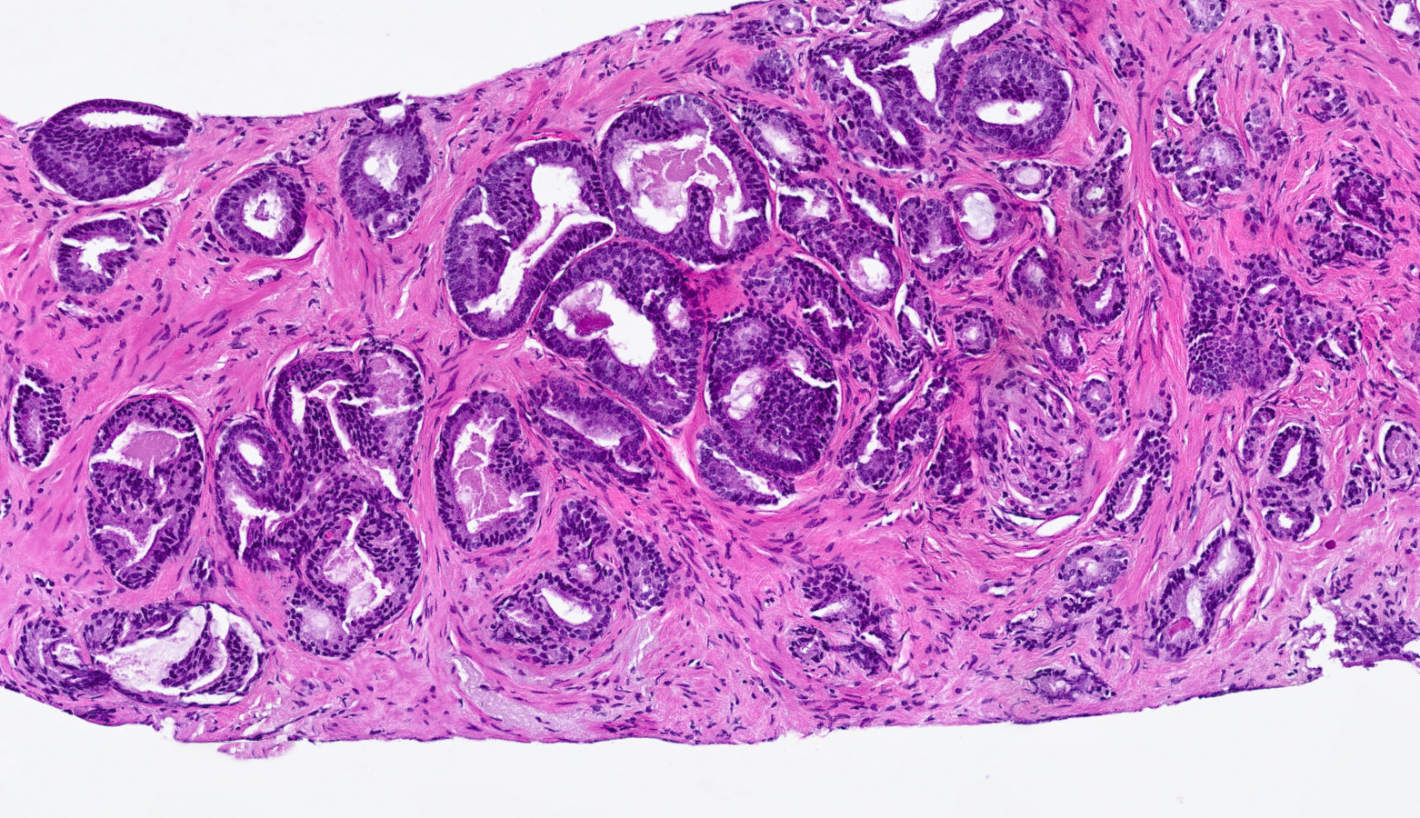
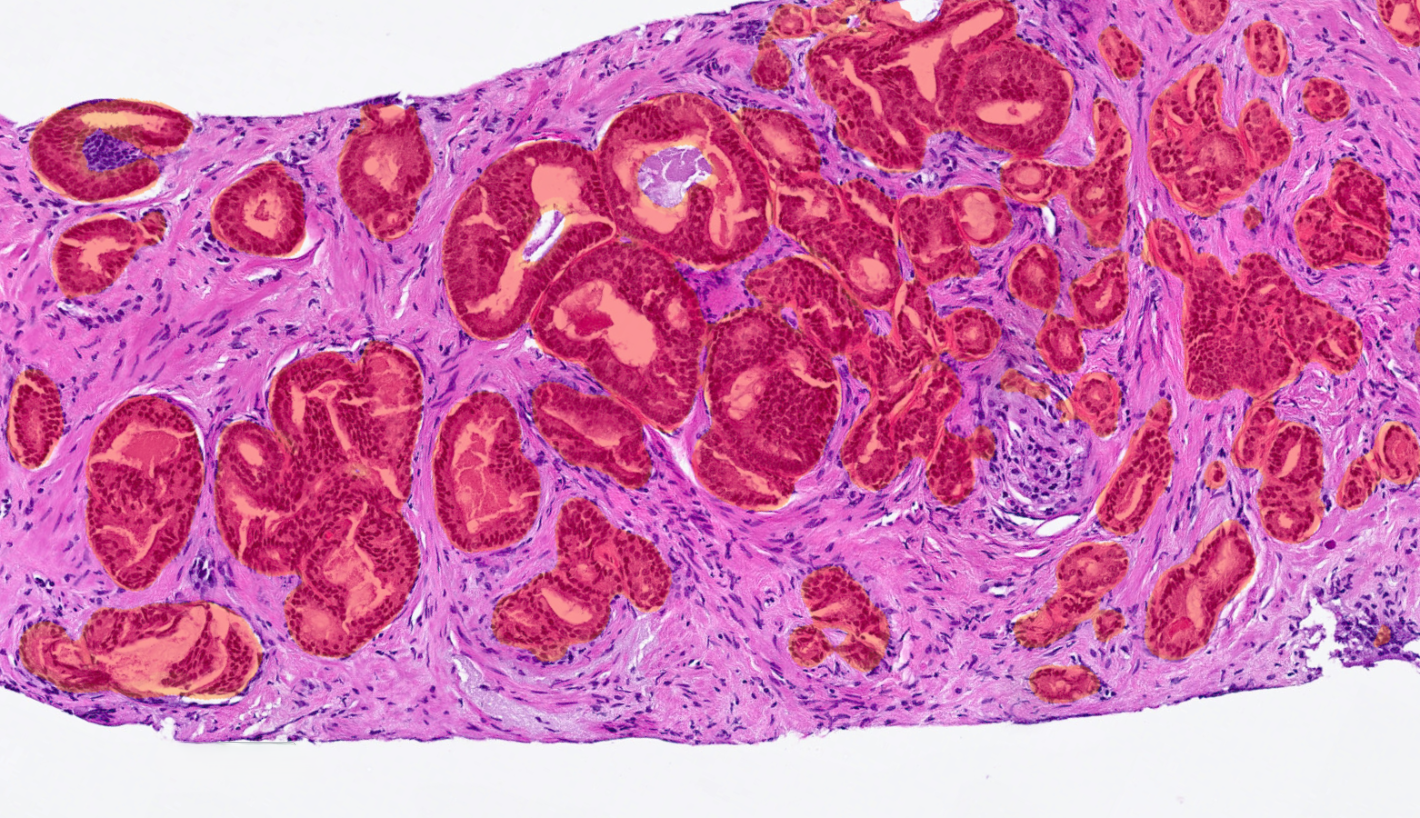

AI for Prostate Tumor Detection and Gleason Grading
Prostate AI is an AI-powered digital pathology analysis tool designed to identify and classify prostate adenocarcinoma in prostate core needle biopsies, helping users standardize Gleason grading for more accurate and reproducible results.
Seamlessly Integrated into HALO AP® for an AI-Enhanced Workflow
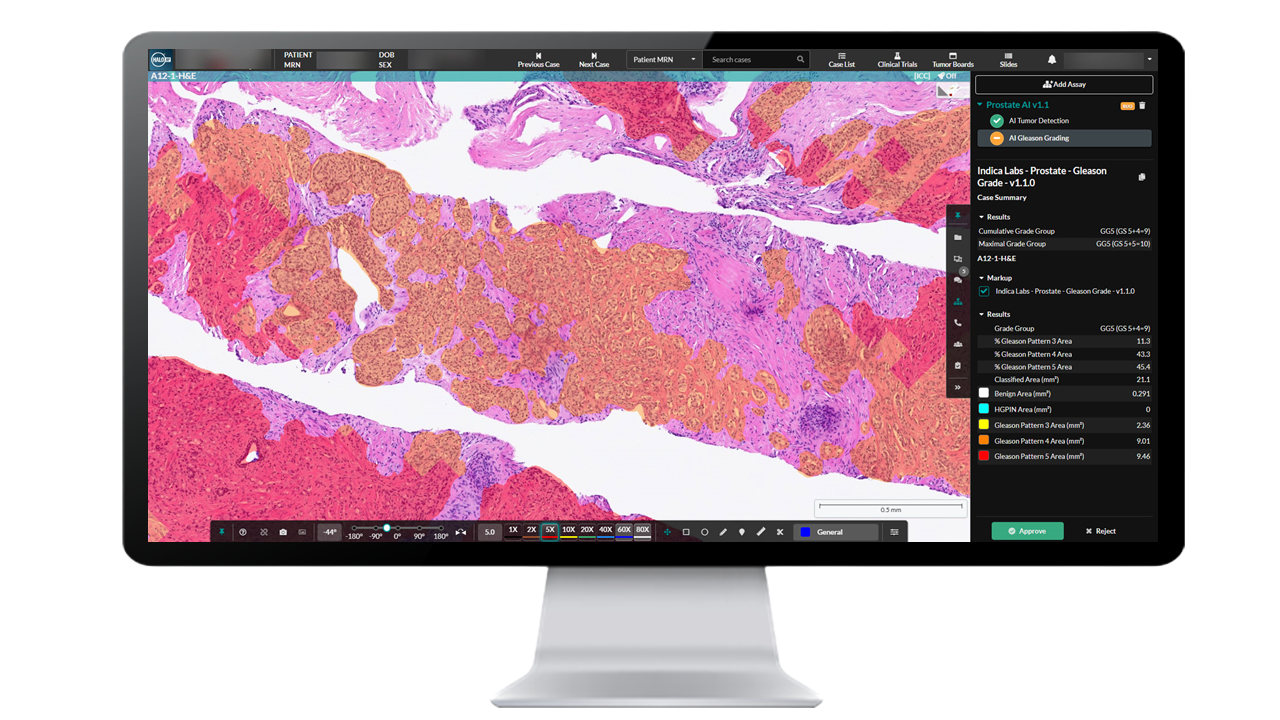
Prostate AI detects and grades prostate adenocarcinoma in core needle biopsies while standardizing Gleason grading on a case and slide level. The prostate algorithm offers a comprehensive analysis of the Gleason grade while displaying an easy-to-read image analysis mask, for an intuitive analytical process.
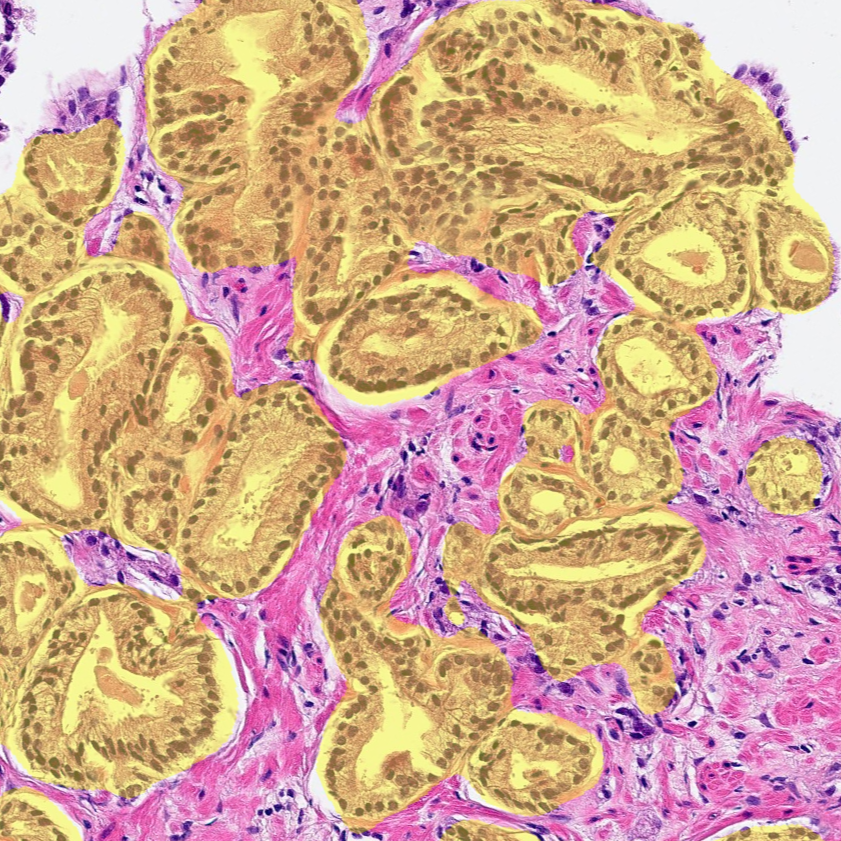
Intended Use
Research Use Only (RUO)
Inputs
H&E whole slide images from prostate core needle biopsies
Key Outputs
- Tumor Detection
- Tumor detected (Yes/No)
- Tumor Area
- Gleason Grading
- Cumulative and Maximal Grade Group
- Grade Group
- Gleason Pattern (% and area)
- Benign Area
- HGPIN Area
File Formats:
- Non-proprietary (JPG, TIF, OME.TIFF)
- Nikon (ND2)
- 3DHistech (MRXS)
- Akoya (QPTIFF, component TIFF)
- Olympus / Evident (VSI)
- Hamamatsu (NDPI, NDPIS)
- Aperio (SVS, AFI)
- Zeiss (CZI)
- Leica (SCN, LIF)
- Ventana (BIF)
- Philips (iSyntax, i2Syntax)
- KFBIO (KFB, KFBF)
- DICOM (DCM*)
*whole-slide images
Detects Prostate Adenocarcinoma Prior to Gleason Grading
AI-powered algorithms detect areas of suspicious prostate adenocarcinoma prior to Gleason grading, helping users identify and classify these tissue regions faster and more accurately.
Early AI-assisted tumor detection supports more accurate and reproducible Gleason grading by improving tumor localization before manual assessment begins.
Pre-screening prostate biopsies with AI can help reduce missed cancerous regions and streamline digital pathology workflows.
Standardizing prostate cancer detection upstream of grading helps laboratories improve efficiency, reduce variability, and support scalable precision oncology workflows.

Seamless Deployment in HALO AP®
Prostate AI is deployed and fully integrated into HALO AP®, the AI-powered, pathologist-driven platform for anatomic pathology workflows from Indica Labs.
Want to Learn More?
Fill out the form below to request a live demo of HALO Prostate AI or to learn more about our other clinical solutions.
You can also drop us an email at info@indicalab.com
Products & Services
Interested in purchasing or learning more about our products and services? Our highly trained application scientists are a couple of clicks away.
Software Maintenance & Support Coverage
Interested in purchasing an SMS plan? We would be happy to give you a quote.
Technical Support
Need technical support? Our IT specialists are here to help.
Prostate AI is for Research Use Only and not intended for clinical diagnostic use. Prostate AI is accessed via the HALO AP® enterprise digital pathology platform.
HALO AP® is CE-IVDR marked for in-vitro diagnostic use in Europe, the UK, and Switzerland. HALO AP® is For Research Use Only in the USA and is not FDA cleared for clinical diagnostic use. In addition, HALO AP® provides built-in compliance with FDA 21 CFR Part 11, HIPAA, and GDPR. WBS-MAR-000002v1
