SlideQC BF is an AI-powered application for H&E- and IHC-stained whole slide images that enables an automated and robust quality control workflow by detecting the most common artifacts generated during the process of slide preparation and scanning including air bubbles, dust/debris, folds, out-of-focus areas, and pen marks. It is seamlessly integrated into the HALO AP® digital pathology platform.
Intended Use
SlideQC BF is an AI-powered quality assurance tool for whole slide images. It assists laboratory personnel with quantification of the most common artifacts generated during slide creation and scanning.
Inputs
H&E- and IHC-stained whole slide images
Key Outputs
A visual mask identifying areas of artifacts and a calculation of area and percentage of slide affected by artifacts.
SlideQC BF was validated on external dataset images scanned by Pannoramic 1000 (HistoQC) and Pannoramic 250 Flash III scanner (Lyon 19) from 3D HISTECH.
Performance by Artifact Type

Validation on external H&E and IHC test sets

SlideQC BF achieved high precision, recall, and F1-score on external test sets of H&E and IHC images.
To learn more about the validation of SlideQC BF, reach out to info@indicalab.com.
File Formats
- Non-proprietary (JPG, TIF, OME. TIFF, DICOM [DCM*])
- Leica (SVS, AFI, SCN, LIF)
- Hamamatsu (NDPI, NDPIS)
- Philips (iSyntax, i2Syntax)
- 3DHistech (MRXS)
- Nikon (ND2)
- Akoya (QPTIFF, component TIFF)
- Olympus / Evident (VSI)
- Zeiss (CZI)
- Ventana (BIF)
- KFBIO (KFB, KFBF)
*whole-slide images

SlideQC BF Brochure
Learn more about the automated quality control and benefits of SlideQC BF.

AI-Assisted Quality Control for Artifact Detection: Deployment in Image Management System with GLP Support
Download our poster to learn how artifact detection with the AI-powered SlideQC BF can advance your quality control pipelines with the GLP support of HALO Link.

An Automated Deep Learning Artifact Detection Tool for Quality Control of Whole-Slide Digital Pathology Images
Explore SlideQC: an AI-based tool which identifies pre-analytic processing errors in whole-slide images for quality control in digital pathology laboratories.

Improve Workflow Efficiency
Providing pathologists with high-quality digital slides expedites report generation and improves workflow efficiency.

Increase Staff Availability
SlideQC BF automates quality control, freeing technicians and staff to work on other laboratory tasks.
Improve Consistency in Lab Operations
SlideQC BF delivers consistency to lab operations through a user-defined threshold that triggers additional review processes.
AI-Powered Quality Control
SlideQC BF automatically analyzes each slide to assess tissue adequacy and detect artifacts, reducing case turnaround times by ensuring only high-quality slides are submitted for pathologist review.


Seamless Deployment in HALO AP®
SlideQC BF is deployed and fully integrated into HALO AP®, the AI-powered, pathologist-driven platform for anatomic pathology workflows from Indica Labs.

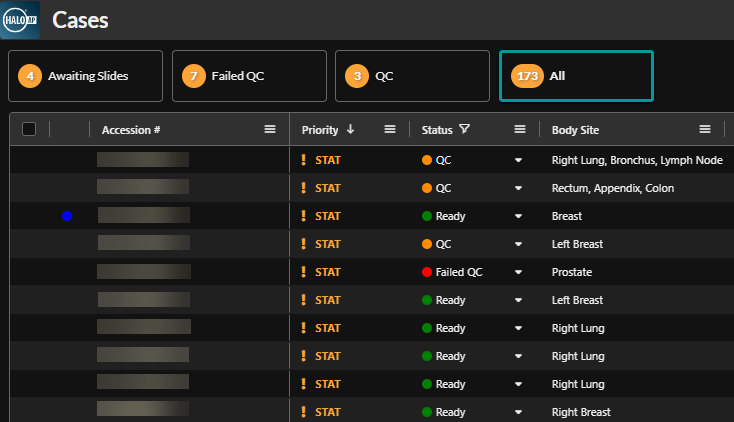
Advanced Workflow Features in HALO AP®
Within HALO AP®, SlideQC BF can automatically tag cases with slides meeting or exceeding a laboratory-defined artifact threshold for further review. Cases can then be triaged into separate worklists for automatic quality control workflows that determine ‘further review’ or ‘ready’ status.
Want to Learn More?
Fill out the form below to request a live demo of SlideQC BF or to learn more about our other clinical solutions.
You can also drop us an email at info@indicalab.com
Products & Services
Interested in purchasing or learning more about our products and services? Our highly trained application scientists are a couple of clicks away.
Software Maintenance & Support Coverage
Interested in purchasing an SMS plan? We would be happy to give you a quote.
Technical Support
Need technical support? Our IT specialists are here to help.
SlideQC BF is intended to be used as a quality control tool for whole slide images and is not regulated as a medical device in the EU/UK. SlideQC BF is for Research Use Only in the USA and is not FDA cleared for clinical diagnostic use. SlideQC BF is accessed via the HALO AP® enterprise digital pathology platform.
HALO AP® is CE-IVDR marked for in-vitro diagnostic use in Europe, the UK, and Switzerland. HALO AP® is For Research Use Only in the USA and is not FDA cleared for clinical diagnostic use. In addition, HALO AP® provides built-in compliance with FDA 21 CFR Part 11, HIPAA, and GDPR.