AI and Digital Pathology in Personalized Medicine: The Past, Present, and Future of Breast Cancer Care


- By Jenna Karpel
- March 5, 2026
Breast Cancer: Tumor Biology as the Foundation of Precision Care
Breast cancer remains a leading cause of cancer-related mortality worldwide, and prior to the advent of precision medicine, treatment options were limited and outcomes were poor. The discovery of predictive, prognostic, and immunologic biomarkers fundamentally transformed breast cancer diagnosis and management, enabling therapies to be tailored to tumor biology instead of primary tumor location alone.
Given the inherent heterogeneity of breast cancer, prognosis and treatment selection are strongly influenced by estrogen receptor (ER), progesterone receptor (PR), HER2 expression, and the Ki67 proliferative index.1 Advancements in molecular biology have further highlighted the variability in tumor behavior and therapeutic response, accelerating the integration of precision medicine into routine care. In parallel, AI-powered digital pathology has emerged as a critical analytical framework, delivering the precision, reproducibility, and scalability required to translate complex biomarker data into confident clinical decisions.
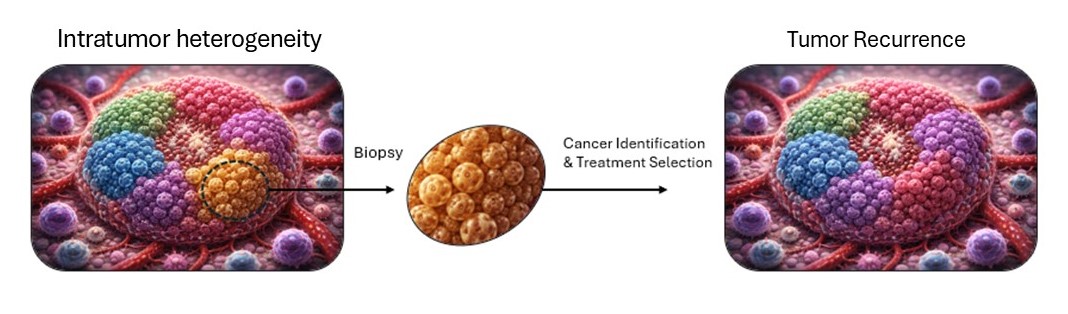
Tumor heterogeneity limits targeted therapy because different cell subpopulations can vary in biomarker expression and drug sensitivity. While treatments may eliminate dominant clones, resistant subclones often persist, driving incomplete responses. This diversity highlights the need for comprehensive profiling and combination strategies that address intratumoral variability.
What Are the Core Biomarkers in Breast Cancer?
ER, PR, HER2, and Ki67 remain the most routinely assessed biomarkers in breast cancer due to their established predictive and prognostic value.
- Predictive biomarkers identify patients who are most likely to benefit from a specific therapy.
- Prognostic biomarkers reflect intrinsic tumor aggressiveness, recurrence risk, and survival outcomes.
ER and PR function as both predictive and prognostic markers as ER positivity is the primary determinant of endocrine therapy benefit, with PR expression serving as a surrogate of intact ER signaling. Loss of PR in ER-positive tumors is associated with diminished endocrine responsiveness and emerging resistance.2
Ki67, a nuclear marker of proliferation, is primarily prognostic, with higher expression correlating with higher histologic grade, increased recurrence risk, and shorter disease-free survival.3
HER2, a transmembrane growth factor receptor, drives proliferation and survival of tumor cells. Historically, HER2 overexpression or gene amplification was associated with aggressive disease biology, endocrine resistance and poor clinical outcomes in the absence of targeted therapy.4
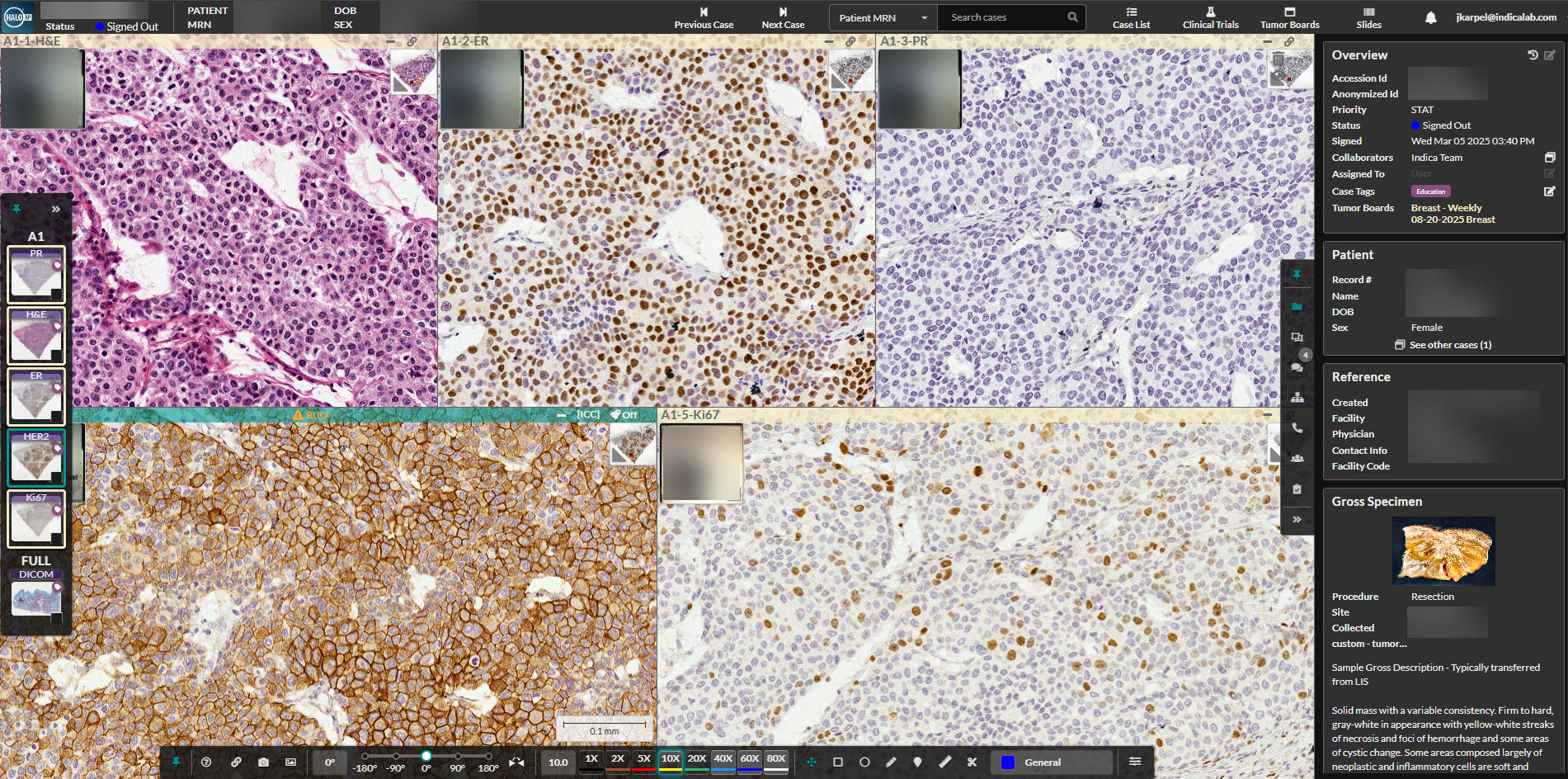
Indica Labs’ in-house developed AI algorithm Breast IHC AI* is shown as a research tool for exploratory breast cancer biomarker detection and quantification launched from their digital pathology platform, HALO AP®. Although the algorithm is not intended for diagnostic, prognostic, or treatment implications, it allows users to analyze all four breast cancer biomarkers simultaneously for a comprehensive analysis of each tumor specimen at a cellular and pixel level.
*For Research Use Only. Not intended for clinical diagnostic use or treatment decision making.
How has the clinical interpretation of HER2 changed?
HER2 is one of the most impactful success stories in precision oncology. First identified as a marker of poor prognosis, it became an ideal target for precision medicine due to both its clinical relevance and its cell surface localization, which enables effective targeting with monoclonal antibodies and antibody-drug conjugates.
For decades, clinical benefit was limited to tumors demonstrating strong HER2 overexpression by immunohistochemistry (IHC) or gene amplification by in situ hybridization. The traditional binary interpretation of HER2 expression as either positive or negative, eligible or ineligible, has been recently challenged by landmark clinical trial data.
Results from DESTINY-Breast04 demonstrated meaningful therapeutic benefit in patients with HER2-low tumors, and emerging evidence suggests a therapeutic response even in HER2-ultralow disease.5 This trial resulted in additional indications for HER2 targeted therapeutics and dramatically expanded the eligible patient population while simultaneously exposing the limitations of traditional, manual biomarker assessment. Detecting subtle differences in HER2 expression that fall below conventional thresholds is not reliably achievable by human interpretation alone, reinforcing the need for advanced digital pathology and AI-driven analysis.
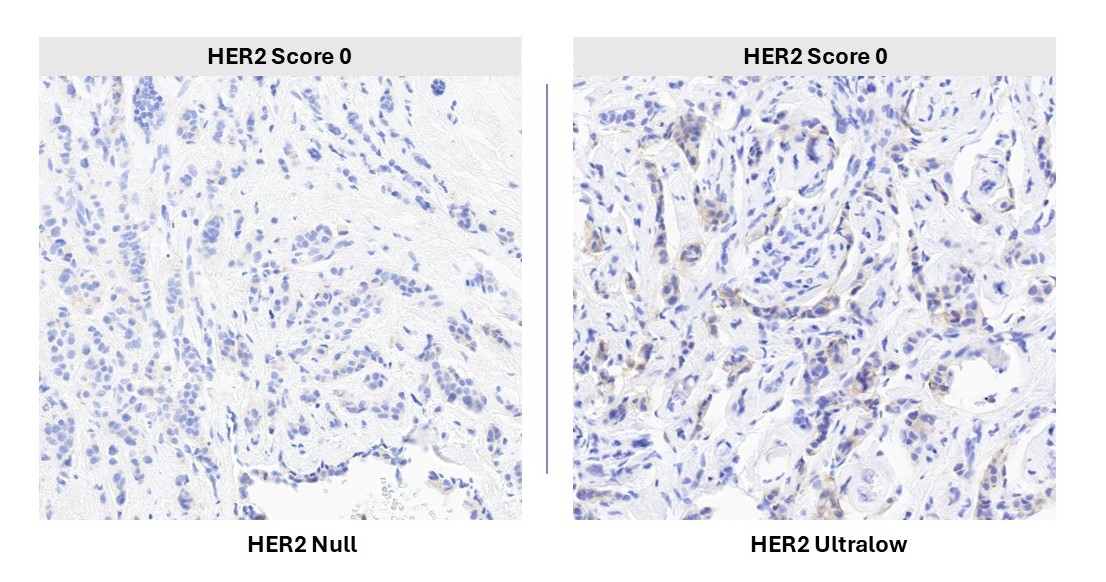
The image shows breast tumor samples analyzed with Indica Labs’ Breast IHC AI (RUO)*. Both specimens are classified as HER2 0; however, the left represents true HER2-null with no detectable staining, while the right demonstrates HER2-ultralow expression (defined by ASCO-CAP as >0% to ≤10% staining). This comparison highlights subtle staining differences, intratumoral heterogeneity, and the narrow expression thresholds described in clinical literature that are associated with targeted therapy eligibility.
* For Research Use Only. Not intended for clinical diagnostic use or treatment decision making.
Why is manual biomarker scoring no longer sufficient?
Long before HER2-low and HER2-ultralow classifications became relevant in clinical practice, manual biomarker scoring was recognized as inherently subjective. Visual estimation of staining intensity and the percentage of positive cells introduces inter- and intra- observer variability, limiting reproducibility and standardization across laboratories and clinical trials.
These challenges are rooted in fundamental neurophysiological constraints as humans are not capable of objectively quantifying subtle color gradients or consistently distinguishing IHC membrane staining intensity differences at scale.6 The downstream consequences are clinically significant, including inconsistent risk stratification, patient misclassification, suboptimal therapy selection, increased cost and toxicity, and diminished confidence in precision medicine.
- Inconsistent risk stratification can lead to overtreatment of patients with low-risk disease, leading to increased toxicity and diminished quality of life. Conversely, patients with high-risk disease could be treated as low-risk patients, leading to preventable disease progression.
- Patient misclassification can lead to the administration of the wrong therapeutic, resulting in adverse reactions and disease left properly untreated.
- Suboptimal therapy selection could result in the patient not receiving the best treatment which could have major impacts on survival outcomes and timeframe eligibility of proper therapeutics.
- The financial burden of precision medicine is substantial, and overtreatment further compounds avoidable healthcare costs for patients.
AI-enabled biomarker quantification directly addresses these challenges by improving analytical precision and consistency while preserving the pathologist’s central interpretive role and adherence to validated clinical standards.
What Pre-Analytical Steps are Required for Reliable AI-Based Biomarker Analysis?
Just as conventional histopathology depends on standardized tissue fixation, section, and staining, AI-driven tissue analysis requires robust pre-analytical digital workflows to ensure analytical validity and reproducibility.
Clinically, ER, PR, HER2, Ki67 and molecular assays are intended to reflect the biology of invasive carcinoma only. From a computational standpoint, AI algorithms are trained and validated exclusively on invasive tumor regions. Inclusion of clinically irrelevant tissue introduces background noise, violates intended use, and degrades analytical accuracy.
For this reason, ductal carcinoma in situ (DCIS) and non-tumor components such as positive tissue microarray (TMA) control cores are identified and excluded from analysis prior to AI-driven biomarker quantification. In this context, a positive TMA refers to an area of strongly positive tissue used for quality control within the lab, but if this positive control is included in the final analysis, it can artificially inflate expression values and push cases across clinically meaningful thresholds.
This workflow reinforces a critical principle that establishes AI to assist, not replace, established laboratory and pathology standards, enabling confident integration into routine clinical workflows.
Why is breast cancer biomarker assessment moving from categories to continuum?
As precision oncology evolves, therapeutic decisions increasingly depend on subtle differences in biomarker expression. This trend is driving a transition from categorical scoring systems to continuous, high-resolution quantification.
Traditional IHC scoring frameworks, such as the HER2 0/1+/2+/3+ system, were developed to simplify manual interpretation in the pre-digital era. While historically essential, this system falls short in the digital era; it defines arbitrary thresholds that impose artificial boundaries, causing category cut offs that have major implications on treatment eligibility, prognosis, and entry into a clinical trial. Additionally, inter-observer variability is particularly pronounced at category boundaries where concordance declines even among expert pathologists.
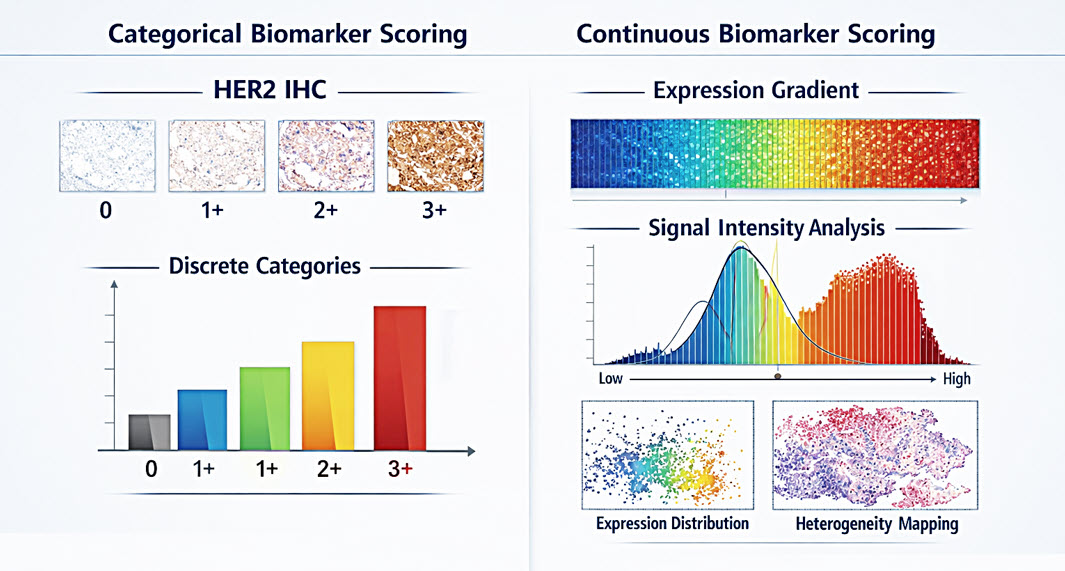
Continuous biomarker scoring powered by digital pathology preserves biologic nuance, reduces subjectivity, and delivers the quantitative precision needed to guide next-generation diagnostics, targeted therapy selection, and the future of precision medicine. Shown for educational and research context only.
How does continuous biomarker scoring improve precision medicine?
Continuous biomarker scoring enables objective, high-resolution quantification of IHC staining intensity at the cellular and pixel level. This approach captures intratumoral heterogeneity and regional variability which is especially important in breast cancer tumors as they are notoriously heterogeneous.
Rather than forcing tumors into discrete categories, continuous methods provide a distribution-based representation of expression that more accurately reflects underlying tumor biology. Continuous scoring is especially important as emerging antibody drug conjugates targeting TROP-2, an exciting actionable target expressed in most breast cancer subtypes like triple negative breast cancer (TNBC), are showing promising efficacy in multiple phase III trials. 7,8 Digital pathology provides the technical infrastructure required to operationalize continuous scoring, including whole-slide imaging, algorithmic intensity measures, and standardized analysis across millions of data points.
What’s next?
As precision oncology continues to advance, cancer diagnostics must move beyond categorical scoring toward continuous, biology-driven measurement. Subtle gradients in biomarker expression, like we see in HER2 and TROP-2, now determine therapeutic eligibility, exposing the limitations of manual interpretation. AI-powered digital pathology enables objective, pixel-level quantification at scale, improving reproducibility, standardization, and access to clinically actionable insights. As emerging therapies increasingly depend on nuanced biomarker expression, digital pathology and AI are no longer complementary technologies, they are foundational to the future of cancer diagnosis and precision care.
Regulatory
Breast IHC AI is For Research Use Only and not intended for clinical diagnostic use. Breast IHC AI is accessed via the HALO AP® enterprise digital pathology platform.
HALO AP® is CE-IVDR marked for in-vitro diagnostic use in Europe, the UK, and Switzerland. HALO AP® is For Research Use Only in the USA and is not FDA cleared for clinical diagnostic use. In addition, HALO AP® provides built-in compliance with FDA 21 CFR Part 11, HIPAA, and GDPR. WBS-MAR-000019v1
References
1.) Li J, Chen Z, Su K, Zeng J. Clinicopathological classification and traditional prognostic indicators of breast cancer. Int J Clin Exp Pathol. 2015;8(7):8500-8505. Published 2015 Jul 1.
2.) Arpino G, Weiss H, Lee AV, et al. Estrogen receptor-positive, progesterone receptor-negative breast cancer: association with growth factor receptor expression and tamoxifen resistance. J Natl Cancer Inst. 2005;97(17):1254-1261. doi:10.1093/jnci/dji249
3.) Chidananda Murthy G. Ki-67 Index and Its Correlation with Clinical and Pathological Variables in Breast Cancer. Indian J Surg Oncol. 2023;14(4):943-948. doi:10.1007/s13193-023-01833-6
4.) Luo Y, Li Q, Fang J, et al. ER+/PR- phenotype exhibits more aggressive biological features and worse outcome compared with ER+/PR+ phenotype in HER2-negative inflammatory breast cancer. Sci Rep. 2024;14(1):197. Published 2024 Jan 2. doi:10.1038/s41598-023-50755-4
5.) Modi S, Jacot W, Yamashita T, et al: Trastuzumab deruxtecan (T-DXd) vs treatment of physician’s choice in patients with HER2-low unresectable and/or metastatic breast cancer: Results of DESTINY-Breast04, a randomized phase 3 study. 2022 ASCO Annual Meeting. Abstract LBA3. Presented June 5, 2022.
6.) Scheel AH, Penault-Llorca F, Hanna W, et al. Physical basis of the ‘magnification rule’ for standardized Immunohistochemical scoring of HER2 in breast and gastric cancer. Diagn Pathol. 2018;13(1):19. Published 2018 Mar 12. doi:10.1186/s13000-018-0696-x
7.) Bardia A, Hurvitz SA, Tolaney SM, et al. Sacituzumab Govitecan in Metastatic Triple-Negative Breast Cancer. N Engl J Med. 2021;384(16):1529-1541. doi:10.1056/NEJMoa2028485
8.) Bardia A, Jhaveri K, Im SA, et al. Datopotamab Deruxtecan Versus Chemotherapy in Previously Treated Inoperable/Metastatic Hormone Receptor-Positive Human Epidermal Growth Factor Receptor 2-Negative Breast Cancer: Primary Results From TROPION-Breast01. J Clin Oncol. 2025;43(3):285-296. doi:10.1200/JCO.24.00920